- Products & Services
OrganRXTM Tissue builds quality, transplantable tissue for organ repair needs. Using the advanced biomimetic OrganRXTM technologies allows for the construction of functional organ tissue for therapeutic discoveries.
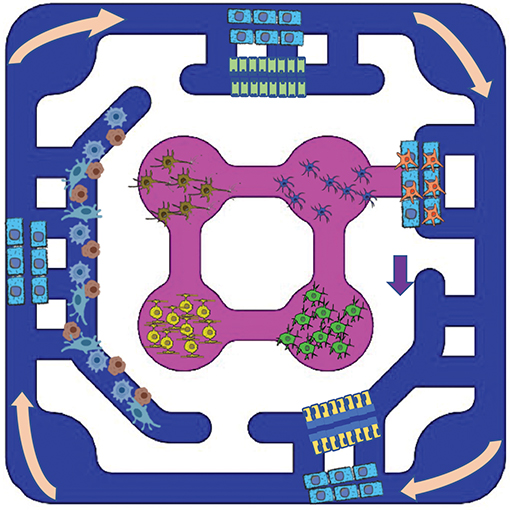
The multi-organ recirculation system (OrganRXTM) provides passive gravity-driven unidirectional shear flow to multi-organ culture. The OrganRXTM plate can accommodate multiple micro-organs such as liver, gut, kidney and brain.The organ plate is an injection molded glass bottom disposable plate. The users can populate the plate with primary cells, cell lines or IPS cells and mature organs by media recirculation. Currently the plate has six multi-organ units.We provide testing of compounds in 2-D and 3-D blood-brain-barrier tissue models. OrganRXTM enables determination of transport characteristics of novel compounds to/from the brain and other organs. - Applications
Applications
OrganRXTM helps to recapitulate human organs with physiological, mechanical and biochemical complexities and their interaction through the exchange of metabolites or signaling molecules and selective transport across the barriers.
Organ Functions
MenuOrgan Models
Menu - ResourcesMenu
- About
- SupportMenu
- Contact Us
Brain-Gut-Kidney-Liver Plate
The administration, distribution, metabolism, and excretion (ADME) of pharmaceutical therapeutics involve multiple organs as the compounds are utilized and processed by various cell types. Drugs targeting the brain are metabolized by the gut and liver and then cleared by the kidney as they flow through the body. Brain-targeting drugs that require direct interaction with the organ need to be able to pass through the blood-brain barrier.
Culturing cell types from multiple organs in a single platform can be difficult due to the cells needing specific, controlled environments for optimal growth. Current culturing systems can be highly controlled but are static, not mimicking the physiological conditions of the organs in vivo. This also limits each cell from reaching in vivo-like morphology and forming tissue due to the lack of cell-to-cell communication.
Biomimetic models like the organ chip provide a three-dimensional microphysiological platform to culture multiple cell types that form organ-like structures. Dynamic fluid flow adds an environmental parameter that promotes cells to perform in vivo functionalities that would otherwise be limited in static, two-dimensional culture systems. The movement of compounds from organ to organ, and even barrier to barrier, can be assessed in an in vitro model that is translatable to in vivo studies.