- Products & Services
OrganRXTM Tissue builds quality, transplantable tissue for organ repair needs. Using the advanced biomimetic OrganRXTM technologies allows for the construction of functional organ tissue for therapeutic discoveries.
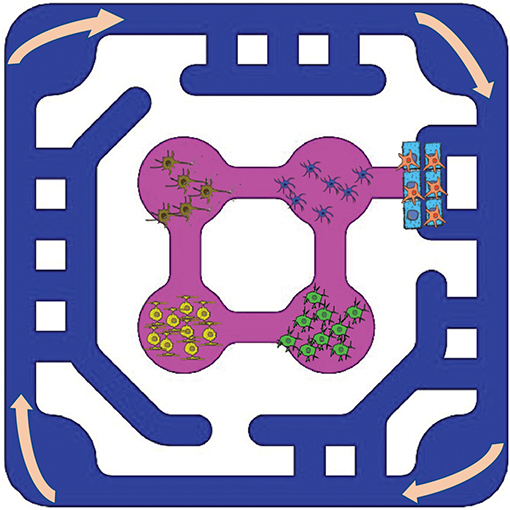
The multi-organ recirculation system (OrganRXTM) provides passive gravity-driven unidirectional shear flow to multi-organ culture. The OrganRXTM plate can accommodate multiple micro-organs such as liver, gut, kidney and brain.The organ plate is an injection molded glass bottom disposable plate. The users can populate the plate with primary cells, cell lines or IPS cells and mature organs by media recirculation. Currently the plate has six multi-organ units.We provide testing of compounds in 2-D and 3-D blood-brain-barrier tissue models. OrganRXTM enables determination of transport characteristics of novel compounds to/from the brain and other organs. - Applications
Applications
OrganRXTM helps to recapitulate human organs with physiological, mechanical and biochemical complexities and their interaction through the exchange of metabolites or signaling molecules and selective transport across the barriers.
Organ Functions
MenuOrgan Models
Menu - ResourcesMenu
- About
- SupportMenu
- Contact Us
BBB Plate
Separating the central nervous system from the blood, the blood-brain barrier (BBB) maintains cerebral homeostasis. The barrier selectively modulates the movement of compounds, molecules, and ions between the blood and the brain. The tight BBB is made up of endothelial cells and fortified by pericytes and astrocytes, and inter- and intra-cellular components are crucial to retaining the barrier’s integrity. Weakened BBB integrity plays a role in a number of pathological conditions that affect the brain and can contribute to neurological diseases like Parkinson’s Disease and other neurodegenerative disorders.
Treating neurological conditions requires drugs to effectively reach molecular targets in the brain, which requires passage through the BBB. Current 2D and transwell models are highly controllable but do not provide physiologically relevant data on barrier integrity and activity comparable to in vivo functionality. Their static states also do not allow cells to grow in in vivo-like microenvironments, limiting cell morphology and function.
Microfluidic, 3-D modeling of the BBB promotes cells to assume in vivo physiology. These models can help study how dysfunction of the barrier can lead to pathological pathways. Additionally, these BBB models can help in drug screening and development as the ability of compounds to pass the barrier is assessed with biological fluid flow.