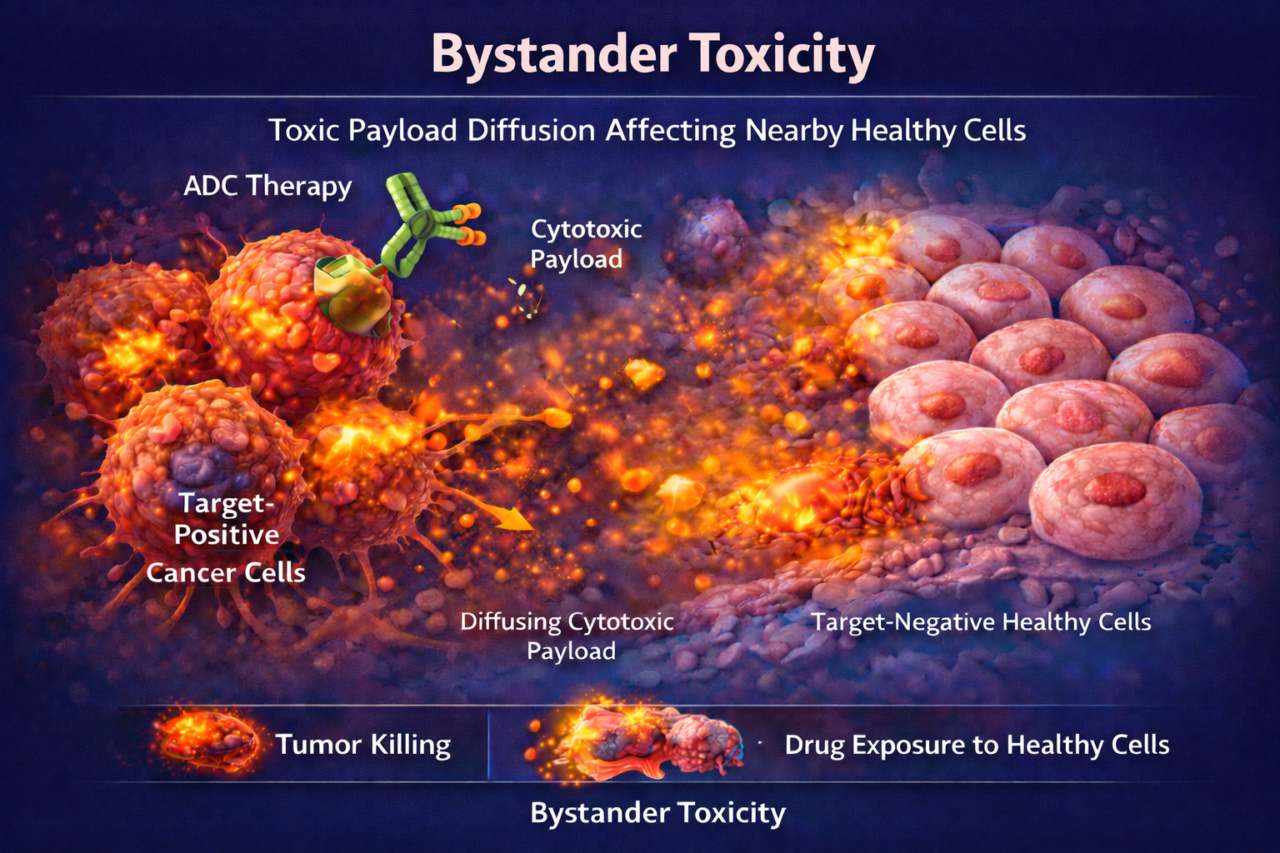
Bystander toxicity arises when cytotoxic payloads from antibody-drug conjugates (ADCs) or other targeted therapies diffuse into adjacent cells that lack the target antigen. For ADCs, a membrane-permeable payload (like MMAE or SN-38) can exit the target cell and kill neighboring cells. While some bystander killing can improve tumor clearance, excessive diffusion risks damaging normal tissue. This is a critical concern for ADC development because the same mechanism that kills tumor cells can harm surrounding healthy cells.
In clinical development, bystander toxicity is difficult to quantify preclinically. Standard 2D cell culture cannot mimic the spatial architecture of tissues, and animals may metabolize ADCs differently. Thus, unpredictable off-target effects often only surface in patient studies. Regulators are aware of cases where ADC payloads caused unexpected liver or lung toxicity in trials, so understanding this phenomenon is essential.
OrganRX™ addresses this gap. For instance, we co-culture target-positive tumor cells and target-negative “bystander” cells in adjoining OrganRX™ channels. After perfusing with an ADC, we measure killing in both compartments. Our models with integrated liver modules could also reveal how metabolites contribute to bystander effects systemically.
Methods and assays: In practice, we use tissue models containing mixed target-expressing and non-expressing cells under flow. We dose the ADC into the recirculation channel and then assess cell viability spatially. Imaging (e.g. fluorescent viability stains) shows if cell death spreads beyond the target cell cluster. We also measure levels of free payload in the media over time by immunoassay. A key assay is comparing effects of cleavable-linker ADCs (which allow bystander diffusion) versus non-cleavable ADC controls.
Biomarkers to validate: Within OrganRX™, we quantify cytotoxicity in both target and non-target cells (e.g. caspase activity, viability dyes). Tracking the concentration of free drug or payload in the medium provides a direct measure of diffusion. Tissue-specific injury markers (e.g. hepatocyte enzymes for liver damage) can indicate organ-level bystander effects. Regulators will expect evidence of how payload release correlates with off-target toxicity; thus, demonstration that a non-cleavable ADC shows lower bystander killing can support safety claims.
FDA perspective: For ADCs, the FDA scrutinizes both the antibody specificity and the payload distribution. Developers must present data on linker stability, payload release kinetics, and off-target cytotoxicity. OrganRX™ Plate data showing restricted payload diffusion (or controlled bystander killing) can help justify safety profiles. Such mechanistic insight can complement in vivo toxicity studies, especially when clinical findings suggest off-target effects.
Limitations: Current studies using OrganRX™ Plate to evaluate bystander toxicity remain in early validation stages. While these platforms allow spatial analysis of drug diffusion and cell injury, validation to fully reproduce the complex metabolic processes that occur in the human body is in validation. Despite these limitations, multiorgan platforms provide a promising framework for mechanistically dissecting bystander toxicity and for guiding the safer design and optimization of antibody–drug conjugates during preclinical development.