OrganRX™ : CRO Services for Biologics
Biopico’s OrganRX™ is a multi-organ microphysiological platform designed to support CRO services for biologics, including antigen-specific CD8+ T cells, bispecific T cell engagers, and antibody-drug conjugates. The platform uses a gravity-driven perfusion system to recirculate media and immune cells through multiple human organ models on a single plate, enabling human-relevant evaluation of tumor–immune interactions, drug metabolism, and tissue-specific toxicity in a format that conventional two-dimensional cultures and many animal models do not capture effectively.
By integrating tumor spheroids, liver organoids, vascular endothelium, gut tissues, and circulating immune cells under flow, OrganRX™ enables simultaneous observation of T cell infiltration, tumor killing, cytokine release, and off-target tissue injury. This makes the platform especially valuable for biologics whose activity is shaped by systemic immune responses, bystander effects, and organ-specific toxicity liabilities.
To demonstrate the utility of OrganRX™ for translational biologics development, Biopico has performed case studies in three clinically relevant categories: CEA-positive antigen-specific CD8+ T cell therapies, the CEA × CD3 bispecific antibody Cibisatamab, and the CEA-targeted antibody-drug conjugate Labetuzumab Govitecan. Across these studies, OrganRX™ has been used to characterize efficacy, safety, cytokine signaling, and tissue injury within a human-relevant multi-organ setting.
Case Study 1: CEA-Positive Antigen-Specific CD8+ T Cells
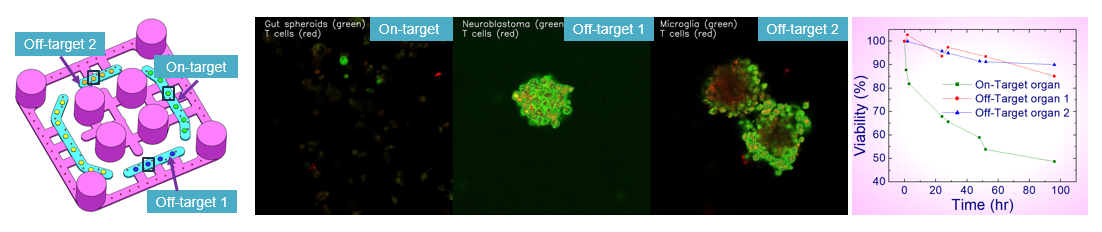
In this study, OrganRX™ was used to evaluate CEA-positive antigen-specific CD8+ T cell therapies in a multi-organ setting designed to assess both tumor cytotoxicity and potential gut or liver toxicity. The study configuration included colorectal cancer spheroids with high CEA expression together with neuroblasto spheroids, microglia spheroids and liver spheroids. Allogeneic CD8+ T cells engineered with a CEA-specific receptor were introduced into circulation at defined effector-to-target ratios.
The system enabled direct monitoring of tumor killing, T cell infiltration, apoptosis induction, and immune activation over several days. Tumor viability was assessed by live/dead imaging and viability assays, while T cell activation markers such as CD69 and CD25 were measured by flow cytometry. Media samples were analyzed for interferon-gamma and tumor necrosis factor-alpha, and gut or liver injury was evaluated through cell death measurements, and liver injury markers such as ALT.
This study design reflects a central challenge in antigen-specific T cell therapy development: balancing efficacy against on-target off-tumor risk. A previous clinical work with CEA-targeted T cells showed the potential for tumor regression but also severe transient colitis, underscoring the importance of including intestinal models when assessing CEA-directed programs. OrganRX™ provides a practical way to evaluate this balance in a human-relevant preclinical system.
Case Study 2: CEA × CD3 Bispecific Antibody: Cibisatamab
In the second case study, OrganRX™ was used to evaluate the CEA × CD3 bispecific antibody Cibisatamab in the presence of human PBMCs or CD8+ T cells and CEA-positive tumor models. Colorectal cancer spheroids with high CEA expression were cultured together with colonic spheroids, neuroblastoma spheroids, microglia spheroids and liver spheroids. Fresh PBMCs or CD8 T cells were added to the circulating media along with a dosing series of Cibisatamab.
The platform enabled characterization of tumor killing kinetics, immune synapse formation, T cell expansion, and cytokine release in one integrated assay. Imaging-based readouts were used to quantify dose-dependent tumor killing, while immunofluorescence assessed CD3–CEA colocalization. Flow cytometry was used to measure T cell activation and expansion, and multiplex cytokine panels evaluated interferon-gamma, interleukin-2, interleukin-6, and tumor necrosis factor-alpha.
This type of study is especially relevant for T cell engagers, where efficacy is closely tied to immune activation but excess cytokine release can limit clinical utility. By simultaneously measuring tumor cytotoxicity and cytokine release thresholds, OrganRX™ helps define the therapeutic window and provides mechanistic data that can inform dose selection and candidate prioritization.
Case Study 3: CEA-Targeted Antibody-Drug Conjugate: Labetuzumab Govitecan
The third case study focused on Labetuzumab Govitecan, an anti-CEACAM5 antibody-drug conjugate carrying the SN-38 payload. OrganRX™ was configured with CEA-positive colorectal tumor spheroids together with liver spheroids, microglia spheroids and neuroblastoma spheroids, payload metabolism, bystander killing, and hepatotoxicity in a multi-organ system.
Labetuzumab Govitecan was introduced into circulation at clinically relevant concentrations, and tumor viability was monitored by imaging and viability assays. Payload delivery and internalization were assessed by immunofluorescence or flow-based methods, and mixed spheroid systems containing both CEA-positive and CEA-negative cells were used to evaluate bystander killing.
This study design addresses a key translational issue for ADC development. While target-dependent cytotoxicity is essential, free or diffusible payload can also damage neighboring cells and healthy tissues. SN-38 is known to be highly potent and associated with gastrointestinal toxicity and neutropenia, making metabolic profiling and bystander assessment particularly important. OrganRX™ provides a system in which these mechanisms can be studied in parallel with tumor response.