Cytokine Release Syndrome
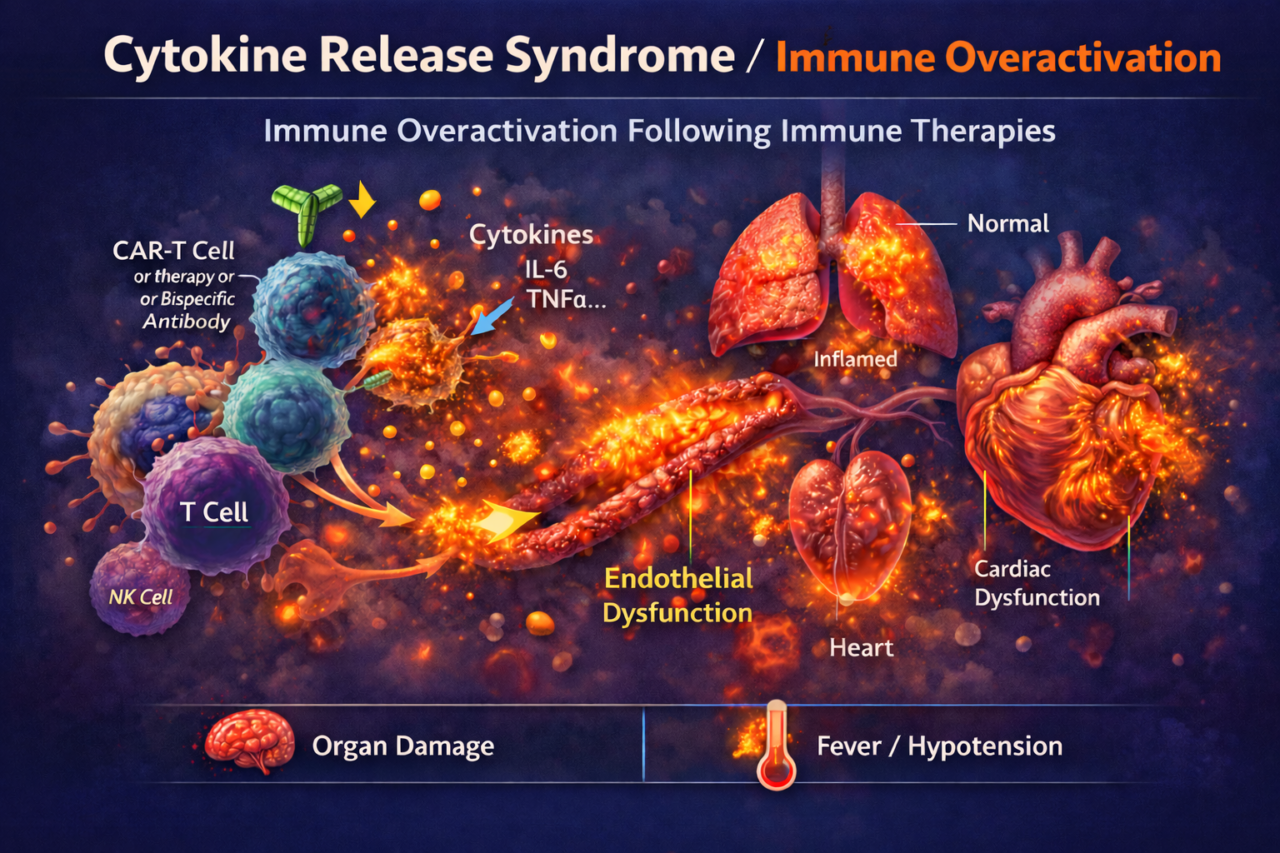
CRS is an acute systemic inflammatory reaction caused by excessive immune cell activation. Therapies that strongly engage T cells or immune pathways can trigger massive release of inflammatory cytokines (IL-6, IFN-γ, TNF-α, etc.) into circulation. This can cause fever, hypotension, coagulopathy, and organ damage. For pharma, CRS is a major safety liability: uncontrolled cytokine storms can lead to patient morbidity or mortality. Strong immune activation also complicates dose finding and may require additional safety precautions in trials.
Clinically, CRS often only appears once the therapy engages the human immune system. Standard in vitro assays (e.g. PBMC cytokine release tests) and animal models poorly predict the full cascade of events. Animals may not replicate human immune cross-talk, and static cell assays lack tissue context. Thus, high CRS risk therapies (like CAR-T or bispecifics) have frequently caused unexpected adverse events in early trials.
OrganRX™ Plate enables the integration of human immune cells with multiple human tissue types under controlled recirculating flow, allowing researchers to observe systemic and organ-specific inflammatory responses. In this platform, circulating PBMCs interact dynamically with tissue compartments, closely mimicking immune–tissue communication in the human body. When the immune cells are exposed to strong inflammatory stimuli such as bacterial endotoxin and interferon signaling, the system generates a systemic cytokine response characterized by elevated inflammatory mediators including tumor necrosis factor alpha, interleukin-6, interleukin-10, monocyte chemoattractant protein-1, and macrophage inflammatory protein-1 alpha. These signals propagate across the interconnected tissues and produce measurable injury in multiple organ compartments, modeling a systemic inflammatory reaction. In contrast, OrganRX™ can distinguish localized organ toxicity from systemic immune activation.
Methods and assays: We run time-course experiments (typically 24–72 hr) in OrganRX™ with circulating PBMCs or purified CD8+ T cells. Immune activation is quantified via cytokine panels (ELISA for IL-6, IFNγ, TNFα, IL-1β, IL-10, chemokines) and by immune-cell markers (CD69, CD25 on T-cells; CD14 on monocytes). Functional readouts indicate organ model impact. For example, on-chip T cells infiltration was visualized, and liver toxicity injury markers ALT/AST and albumin are measured. Comparing treated vs. control chips (no drug, or static vs. flow) reveals CRS effects.
Biomarkers to validate: Key biomarkers include inflammatory cytokines (IL-6, TNFα, IFNγ, IL-1β) and chemokines, which indicate CRS magnitude. Immune-cell activation markers (CD69, CD25) and proliferation rates also reflect overactivation. Tissue (e.g. ALT/AST for liver) can be measured. FDA emphasizes monitoring IL-6 and other cytokines in CAR-T trials, so aligning chip data with such markers is valuable for regulatory discussions.
FDA perspective: CRS is a known concern for immunotherapies, and FDA expects preclinical evaluation of cytokine release risk. Data on cytokine profiles, activation thresholds, and downstream organ effects can guide safety monitoring. Human-chip data documenting cytokine kinetics and organ responses can help support dose selection and risk mitigation plans in IND/CTA submissions.
Limitations: Current studies focus on innate and T-cell responses in the short term (weeks). They may not model full adaptive immunity (no vaccine or memory phases). Immune cell exhaustion and longer-term regulation are not captured. Also, models may not include all key immune cell types (e.g. B cells, neutrophils) equally. Despite this, they offer important early-warning data on hyperinflammation that static assays miss.