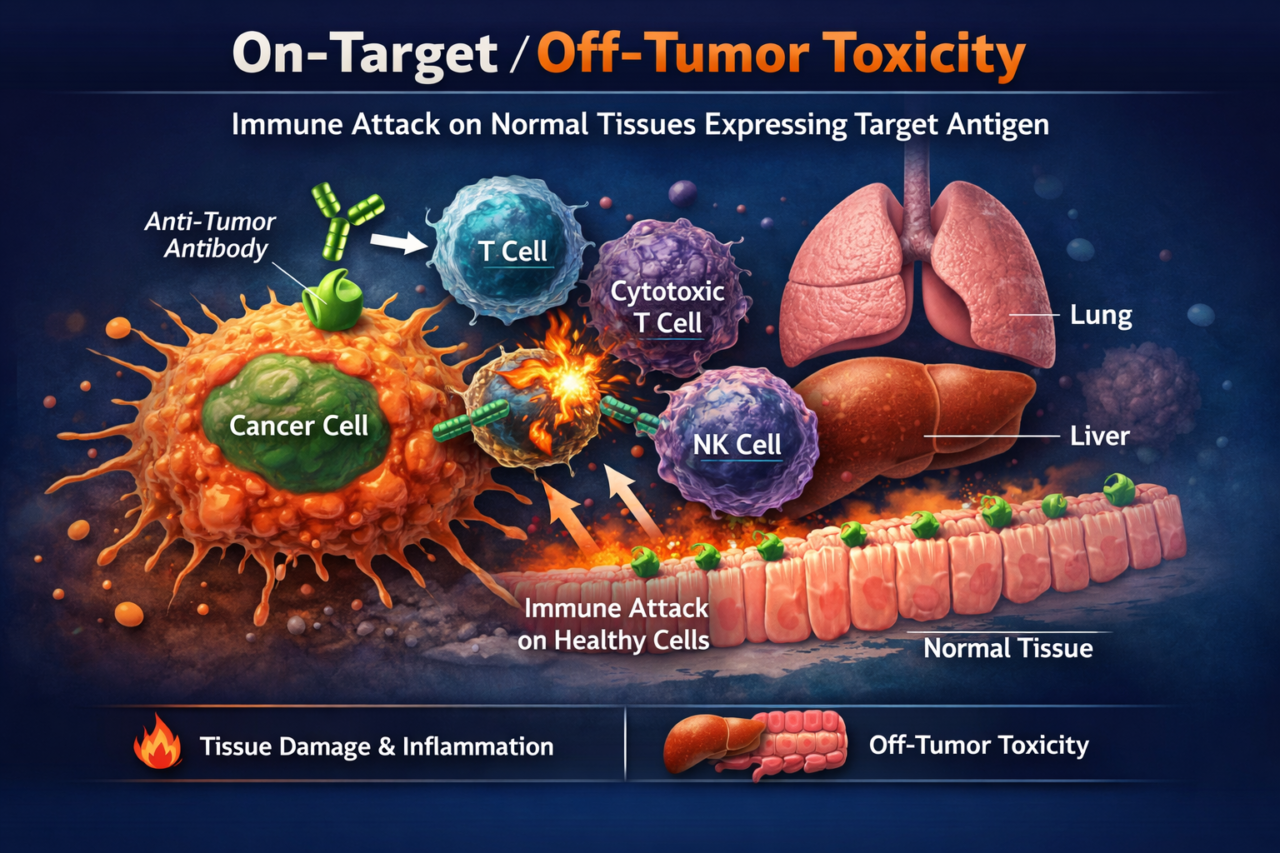
On-Target/Off-Tumor Toxicity
On-target/off-tumor toxicity occurs when a biologic drug correctly binds its target antigen, but that antigen is also expressed in healthy tissues at lower levels. Immune-engaging antibodies or bispecifics then trigger immune-mediated killing of those normal cells. This risk is especially critical for highly cytotoxic therapies. For pharma developers, on-target toxicity directly constrains the safety margin: if vital organs (e.g. lung, gut, kidney) express the target, the drug dose may need to be lowered to avoid organ damage, potentially compromising efficacy. Late-stage discovery of such toxicity can halt development, leading to trial failures and high costs.
Traditional models often fail to predict this risk. Animal species may not express the human target antigen in the same pattern, or the antibody may not bind animal antigen at all. Immune system differences mean animal immune responses may not mirror humans. As a result, on-target toxicities often only become apparent in first-in-human trials.
Multiorgan MPS help by directly using human tissues and immune cells. For example, in Lung-model or Intestine-model seeded with primary human lung or gut epithelium and endothelium, plus flowing human PBMCs, when treated with tumor-targeting bispecific antibodies, these models could reveal precisely which tissues suffer immune attack. For example, in a Lung-model, a high-affinity FOLR1 bispecific induce significant alveolar epithelial apoptosis and CD8^+ T-cell activation (CD69 upregulation). Critically, this will reproduce the on-target lung toxicity seen in non-human primates. Further, in a Colon-model, anti-CEA bispecifics cause dose-dependent immune cell adhesion and killing only where target expression is high. These models also enabled comparison against controls: non-targeting bispecifics (CD3-only) causes no effects, confirming specificity.
Methods and assays: We culture spheroids/organoids or primary cell layers in OrganRX™ Plate with perfused channels. Tissues are matured (~5–10 days) then PBMCs (1×10^6) are introduced along with the biologic drug. We measure tissue injury by cell-death assays (e.g. caspase-3/7 staining, LDH release) and immune activation by flow cytometry (T-cell CD69, CD107a) and cytokine assays. In situ imaging captures immune-cell infiltration. By including multiple tissues in one circuit, we observe off-tumor effects across organs concurrently.
Biomarkers to validate: We focus on indicators of tissue injury (e.g. released LDH, apoptotic markers) and immune activation (CD69^+ T-cells, cytokines IFNγ, TNFα, IL-6, granzyme B). Organ-specific damage markers (e.g. lung surfactant proteins, intestinal tight-junction disruption) can also be monitored. FDA guidance emphasizes demonstrating tissue cross-reactivity; our human data on dose-response and tissue injury patterns directly support safety assessment.
FDA perspective: Regulators consider tissue cross-reactivity a high safety priority for biologics. They expect developers to assess antigen expression in human tissues and characterize potential toxicity mechanisms preclinically. While GLP animal studies are mandatory, agencies recognize their limitations for human-specific targets. Human organ-chip data showing target-dependent injury can strengthen a regulatory submission by clarifying risk and informing clinical dose strategy.
Limitations: Current organ-chip models are short-term (days to weeks), so they best capture acute toxicity. Currently, efforts are underway to model long-term immune memory or chronic inflammation. Donor variability in primary cells can affect reproducibility, requiring multiple replicates. Also, not all organ systems (e.g. nervous or endocrine) are yet incorporated together. Despite these limits, organ-chips provide mechanistic insights into tissue-specific toxicity that animal tests may miss.