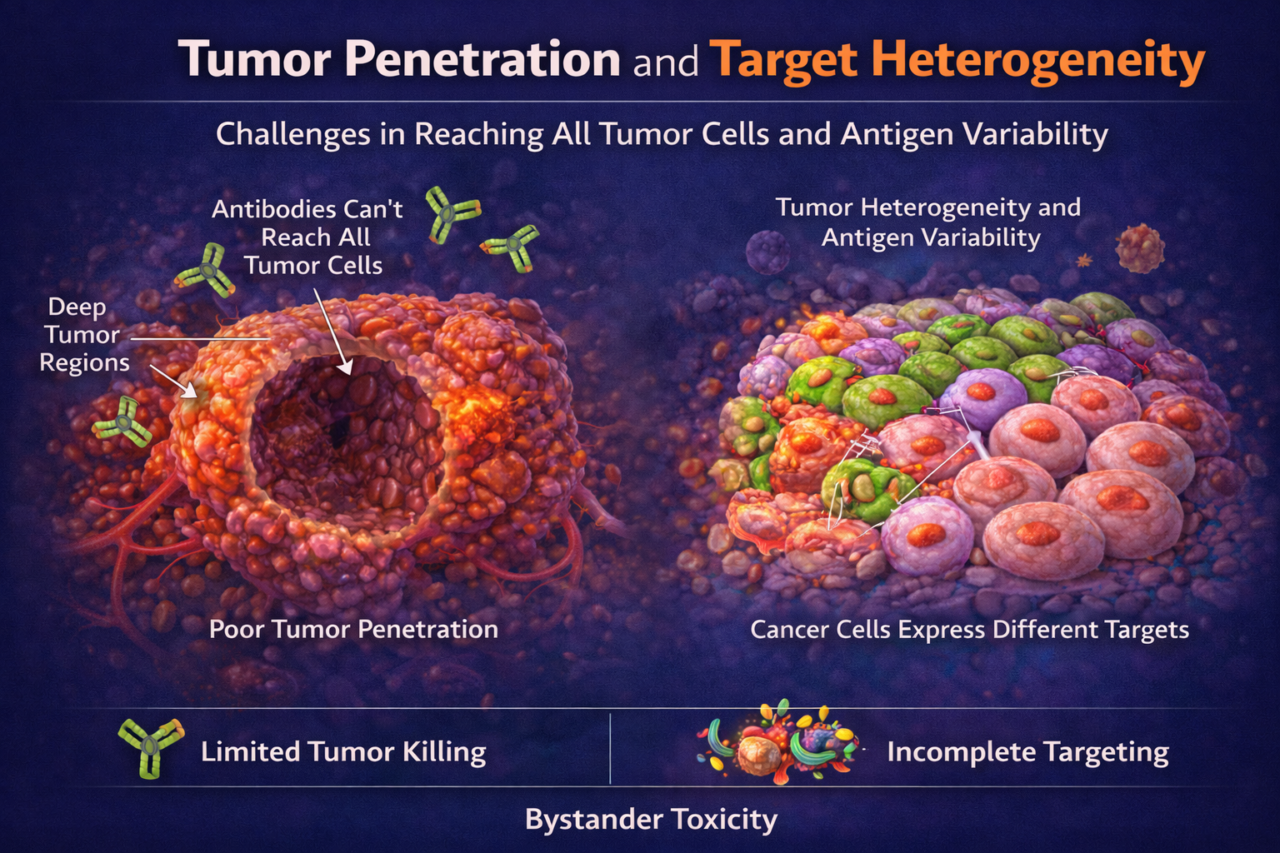
Poor Tumor Penetration / Target Heterogeneity
Large biologic drugs (antibodies, ADCs, cytokines) often fail to reach all tumor cells in solid malignancies. Dense extracellular matrix and abnormal vasculature impede drug penetration. Additionally, tumors are heterogeneous: some cells express high levels of the target antigen while others express none or very little. These factors can allow subpopulations of cancer cells to evade treatment, leading to relapse. Clinically, inadequate tumor penetration and target heterogeneity have been implicated in mixed responses to therapy and treatment resistance.
Conventional preclinical models inadequately predict these challenges. 2D cultures and spheroids do not fully capture tumor physical barriers. Animal models may not mirror human tumor microenvironments. Therefore, patient responses often diverge from expectations. Regulators understand that limited distribution or heterogeneous expression can influence efficacy, so demonstrating coverage of the tumor is important.
OrganRX™ Plate can model these phenomena by integrating 3D tumor tissues with recirculation flow. For example, tumor spheroids or organoids grown within an OrganRX™ Plate under flow with labeled biologic can image its distribution. This approach reveals penetration depth and gradients. Co-culturing stromal cells (fibroblasts, MSCs) can mimic the extracellular matrix that slows diffusion. Including fibroblasts and immune cells can illustrate the impact of heterogeneity in immune therapy response to better match patients.
Methods and assays: Tumor penetration studies use fluorescent drugs. Confocal imaging measures signal intensity versus depth at various time points. Viability assays (e.g. live/dead staining) show which tumor cells survive. PK/PD assays can be run by sampling media for drug concentration over time to inform mathematical diffusion models. Additionally, chips can stratify tumor regions by target expression: for example, co-culturing two tumor organoids with different marker expression levels to test if one protects the other from therapy.
Biomarkers to validate: The key output is the distribution profile of the therapeutic agent, often visualized by fluorescent labeling. Biomarkers include quantitative imaging of antibody or drug within tumor masses, and downstream effects such as target receptor occupancy (if measurable). Tumor cell viability markers (annexin V, caspase-3) across the tissue highlight resistant pockets. Regulators will expect rationale for patient selection (e.g. biomarkers of target expression) and an understanding of how drug dose relates to intratumoral exposure; organ-chip data on distribution can inform these considerations.
FDA perspective: The FDA recognizes that limited tumor exposure and heterogeneous targets can compromise efficacy. While there is no specific regulatory requirement for penetration data, demonstrating mechanistic understanding can strengthen efficacy claims. For example, showing that an ADC fully permeates tumor tissue at clinically relevant concentrations supports its intended effect. Guidance documents suggest using all available evidence to predict clinical PK/PD; organ-chip penetration data could complement animal and clinical biopsy data in IND/CTA submissions.
Limitations: Tumor models are still emerging and often simplify the complexity of real tumors. They may lack the full immune context or the slow interstitial transport seen in vivo. Many models currently focus on short-term penetration (days), so long-term accumulation or tumor remodeling effects are not captured. Despite these constraints, these systems represent an important step toward understanding and optimizing drug delivery to solid tumors.